Mars Odyssey
NASA’s 2001 Mars Odyssey mission created the first global map of chemical elements and minerals that make up the Martian surface.

Key Facts
Launch Date: April 7, 2000, 11:02 am EST
Launch site: Cape Canaveral Air Force Station, Florida
Launch Vehicle: Delta II 7925
Mars Orbit Insertion: Oct. 24, 2001
Mission Status: Currently Operating

Meet Mars Odyssey
Tech Specs
Dimensions | 7.2 feet (2.2 meters) long 5.6 feet (1.7 meters) tall 8.5 feet (2.6 meters) wide |
Total Weight | 1,598.4 pounds (725.0 kilograms) |
Command and Data Handling Subsystem | The heart of this subsystem is a RAD6000 computer, a radiation-hardened version of the PowerPC chip used on most models of Macintosh computers. With 128 megabytes of random access memory (RAM) and three megabytes of non-volatile memory, which allows the system to maintain data even without power, the subsystem runs Odyssey's flight software and controls the spacecraft through interface electronics. The entire command and data handling subsystem weighs 24.5 pounds (11.1 kilograms). |
Power | Electrical power subsystem weighs 189.6 pounds (86.0 kilograms). |
Propulsion | Uses hydrazine propellant with nitrogen tetroxide as an oxidizer, produces a minimum thrust of 144 pounds of force (65.3 kilograms) of force. Each of the four thrusters used for attitude control produces a thrust of 0.2 pound of force (0.1 kilogram) of force. Four 5.0-pound-force (2.3-kilogram-force) thrusters are used for turning the spacecraft. The entire propulsion subsystem weighs 109.6 pounds (49.7 kilograms). |
Communications | Odyssey's telecommunications subsystem is composed of both a radio system operating in the X-band microwave frequency range and a system that operates in the ultra high frequency (UHF) range. The X-band system is used for communications between Earth and the orbiter, while the UHF system is used for communications between Odyssey and any landers present on the Martian surface at any given time. The telecommunication subsystem weighs 52.7 pounds (23.9 kilograms). |
Science
2001 Mars Odyssey has contributed numerous science results supporting the Mars Exploration Program’s overall strategy of “Following the Water.” It has mapped the amount and distribution of chemical elements and minerals that make up the Martian surface. Maps of hydrogen distribution led scientists to discover vast amounts of water ice in the polar regions buried just beneath the surface. Odyssey has also recorded the radiation environment in low Mars orbit to determine the radiation-related risk to any future human explorers who may one day go to Mars.

Science Objectives
To contribute to the four Mars Exploration Program science goals, Mars Odyssey has the following science objectives:
- Globally map the elemental composition of the surface.
- Determine the abundance of hydrogen in the shallow subsurface.
- Acquire high spatial and spectral resolution images of the surface mineralogy.
- Provide information on the morphology of the Martian surface.
- Characterize the Martian near-space radiation environment as related to radiation-induced risk to human explorers.
Science Instruments

Thermal Emission Imaging System
The Thermal Emission Imaging System (THEMIS) views the visible and infrared reflections from the Martian surface to make a map showing the location of different mineral concentrations and their relationship to various landforms.
By looking at the visible and infrared parts of the spectrum, THEMIS is determining the distribution of minerals on the surface of Mars and helping scientists understand how the mineralogy of the planet relates to the landforms.
Tech Specs
Main Job | To determine the distribution of minerals on the surface of Mars |
Location | On the body of the orbiter |
Mass | 24.7 pounds (11.2 kilograms) |
Power | 14 watts |
Data Return | IR Imager: Instantaneous internal rate of 1.17 Mbits/sec. Data rate to spacecraft after real-time compression is 0.6 Mbits/sec. Visible Imager: Instantaneous internal rate of up to 6.2 Mbits/sec. Data rate to spacecraft is <1.0 Mbits/sec. |
Color Quality | 5 visual bands: 0.425 microns, 0.540 microns, 0.654 microns, 0.749 microns, 0.860 microns. 10 infrared bands: 6.78 microns (used twice), 7.93 microns, 8.56 microns, 9.35 microns, 10.21 microns, 11.04 microns, 11.79 microns, 12.57 microns, 14.88 microns. |
Image Size | Visible camera has a 1024 x 1024 pixel array with 5 filters. IR focal plane has 320 cross-track pixels and 240 down-track pixels covered by 10˜1-μm-bandwidth strip filters in nine different wavelengths. |
Image Resolution | Visual images: 59 feet (18 meters) per pixel Infrared images: 328 feet (100 meters) per pixel |
Focal Length | ~7.8 inch (20 centimeter) effective focal length |
Focal Ratio | f/1.6 |
Field of View | IR Imager: 4.6 degree (80 mrad) cross-track by 3.5 degree (60 mrad) down-track FOV with a 0.25 mrad (100 m) IFOV at nadir. Visible Imager: 2.66 degree (46.4 mrad) cross-track by 2.64 degree (46.1 mrad) down-track FOV with 0.045 mrad (18 m) IFOV in 1024 x 1024 pixels at nadir. The two imagers are spatially bore-sighted. |
TES vs. THEMIS
In the infrared spectrum, the instrument uses 9 spectral bands to help detect minerals within the Martian terrain. These spectral bands, similar to ranges of colors, can obtain the signatures (spectral "fingerprints") of particular types of geological materials. Minerals, such as carbonates, silicates, hydroxides, sulfates, hydrothermal silica, oxides and phosphates, all show up as different colors in the infrared spectrum. This multi-spectral method allows researchers to detect in particular the presence of minerals that form in water and to understand those minerals in their proper geological context. THEMIS' infrared capabilities have significantly improved the data from TES, a similar instrument on Mars Global Surveyor.
The instrument's multi-spectral approach also provides data on localized deposits associated with hydrothermal and subsurface water and enables 328-foot (100-meter) images of Martian terrain to be captured in each pixel, or single point, of every image.
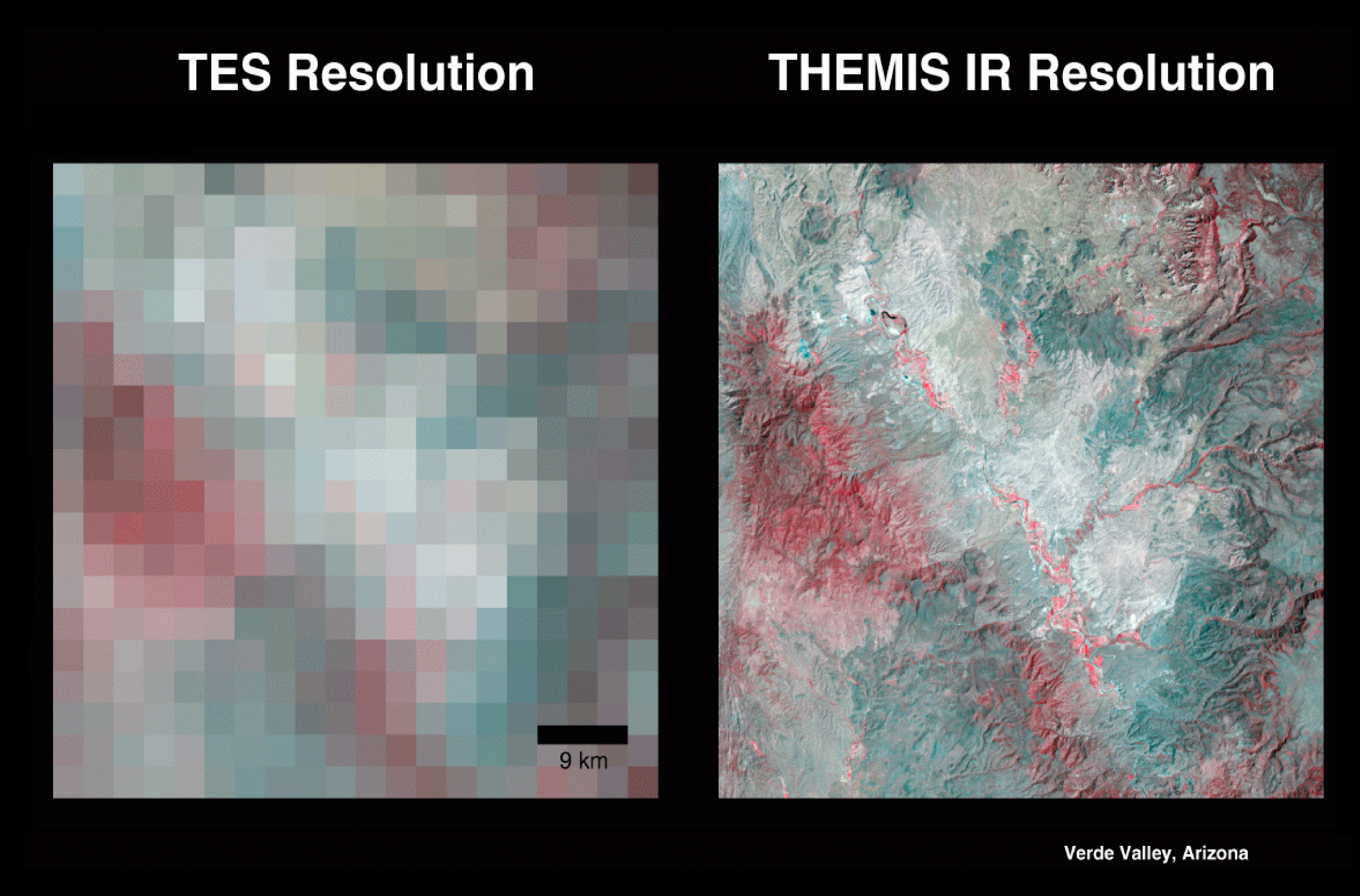
ASTER, an Earth-orbiting instrument on the Terra spacecraft, has used a similar approach to map the distribution of minerals here on Earth.
Variations in the thermal infrared "color" in the right-hand image are due to differences in the kinds of minerals that make up rocks and soil. In the visible part of the spectrum that our eyes can see (left-hand image), it would not be apparent what minerals are present.
How THEMIS Works in the Visible
The part of the imaging system that takes pictures in visible light is able to show objects about as big as a semi-truck. This resolution helps fill in the gap between large-scale geological images from the Viking orbiters in the 1970s and the very high-resolution images from the currently orbiting Mars Global Surveyor.
For more information, visit the THEMIS instrument site at Arizona State University.

Gamma Ray Spectrometer
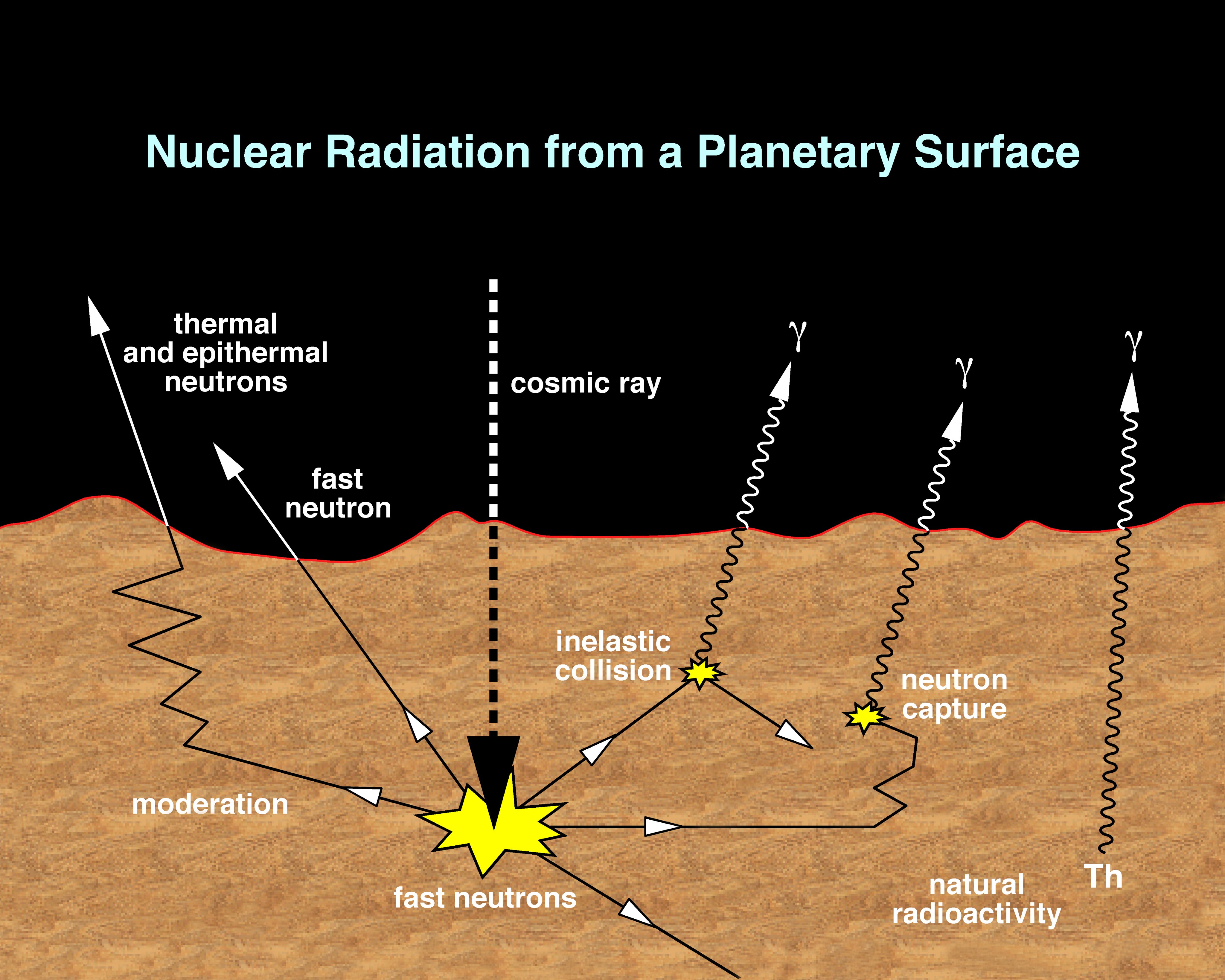
The Gamma Ray Spectrometer (GRS) measures the abundance and distribution of many elements of the periodic table, including hydrogen, silicon, iron, potassium, thorium, and chlorine. Knowing what elements are at or near the surface gives detailed information about how Mars has changed over time. To determine the elemental makeup of the Martian surface, the experiment uses a gamma ray spectrometer and two neutron detectors.
Tech Specs
Main Job | To determine the elemental makeup of the Martian surface |
Location | On the body of the orbiter; gamma sensor head on 20-foot (6.2-meter) boom. |
Mass | 67.2 pounds (30.5 kilograms) |
Power | 32 watts |
Size | Gamma Sensor (GS): 18.4 inches (46.8 centimeters) by 21.0 inches (53.4 centimeters) by 23.8 inches (60.4 centimeters) Neutron Spectrometer (NS): 6.8 inches (17.3 centimeters) by 5.7 inches (14.4 centimeters) by 12.4 inches (31.4 centimeters) High-Energy Neutron Detector (HEND): 11.9 inches (30.3 centimeters) by 9.8 inches (24.8 centimeters) by 9.5 inches (24.2 centimeters) |
Data Return | Collects a new spectrum approximately every 20 seconds, 360 times per orbit. |
Capability | Gamma Sensor (GS): Strikes from high-energy ionizing photons or charged particles are amplified, measured and digitally converted into one of 16,384 (214) channels, or bins. Neutron Spectrometer (NS): Detects neutrons in three energy bands: thermal, epithermal, and fast. High-Energy Neutron Detector (HEND): Neutrons are detected in thermal (0.01 eV - 1 eV), epithermal (1 eV - 1 keV), fast (1 keV - 1 MeV), and high-energy (1 MeV - 10 MeV) energy ranges. |
Spatial Resolution | 186 miles (300 kilometers) |
How GRS Works
When exposed to cosmic rays (charged particles in space that come from the stars, including our Sun), chemical elements in soils and rocks emit uniquely identifiable signatures of energy in the form of gamma rays. The gamma ray spectrometer looks at these signatures, or energies, coming from the elements present in the Martian soil. By measuring gamma rays coming from the Martian surface, it is possible to calculate how abundant various elements are and how they are distributed around the planet's surface.
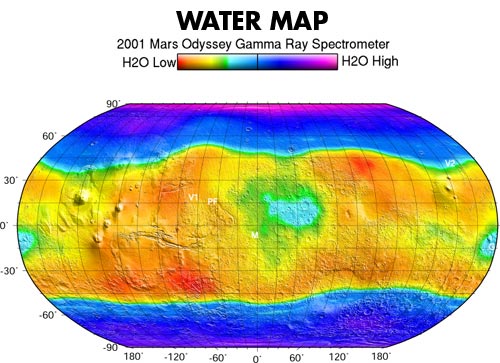
Gamma rays, emitted from the nuclei of atoms, show up as sharp emission lines on the instrument's spectrum. While the energy represented in these emissions determines which elements are present, the intensity of the spectrum reveals the element's concentrations. The spectrometer has added significantly to the growing understanding of the origin and evolution of Mars and the processes shaping it today and in the past.
How GRS Helps Detect Water
By measuring neutrons, it is possible to calculate the abundance of hydrogen on Mars, thus inferring the presence of water. The neutron detectors are sensitive to concentrations of hydrogen in the upper meter of the surface. Like a virtual shovel "digging into" the surface, the spectrometer allows scientists to peer into this shallow subsurface of Mars and measures the amount of hydrogen that exists there. Since hydrogen is most likely present in the form of water ice, the spectrometer is able to measure directly the amount of permanent ground ice and how it changes with the seasons.
Mars Radiation Environment Experiment
Led by NASA's Johnson Space Center, this science investigation characterized aspects of the radiation environment both on the way to Mars and in the Martian orbit.
Since space radiation presents an extreme hazard to crews of interplanetary missions, the experiment predicted anticipated radiation doses that would be experienced by future astronauts and is helping to determine possible effects of martian radiation on human beings.
Mars exploration space radiation comes from cosmic rays emitted by our local star, the Sun, and from stars beyond our solar system as well. Space radiation can trigger cancer and cause damage to the central nervous system. Similar instruments were flown on the space shuttles and are also flown on the International Space Station (ISS), but before Odyssey none had ever flown outside of Earth's protective magnetosphere, which blocks much of this radiation from reaching the surface of our planet.

How the Instrument Works
A spectrometer inside the instrument measured the energy from these radiation sources.
The instrument, with a 68-degree field of view, collected data during Odyssey's cruise from Earth to Mars. It stored large amounts of data for downlink whenever possible, and operated in orbit around Mars until a large solar event bombarded the Odyssey spacecraft on October 28, 2003. MARIE has been unable to collect data since that time, and engineers believe the most likely cause is that a computer chip was damaged by a solar particle smashing into the MARIE computer board.
Tech Specs
Main Job | To help provide a context for high-resolution analysis of key spots on Mars provided by HiRISE and CRISM. |
Location | On the body of the orbiter |
Mass | 7.3 pounds (3.3 kilograms) |
Power | 7 watts |
Size | 11.6 inches (29.4 centimeters) by 9.1 inches (23.2 centimeters) by 4.3 inches (10.8 centimeters) |
Data Return | Data is saved on 60 Mb of flash memory and transferred at <8 Mbits per day over an RS-422 low speed data line. |
Capability | Measures particles in the range of 15 MeV to 500 MeV/nucleon. |
Field of View | 68 degrees |








